Abstract
Background: Blastic plasmacytoid dendritic cell neoplasm (BPDCN) is a rare hematologic neoplasm involving skin lesions and disseminated disease into bone marrow, peripheral blood, and lymph nodes, characterized by poor clinical outcomes and no standard therapeutic approaches. BPDCN is characterized by the malignant proliferation of precursor plasmacytoid dendritic cells (pDCs). It is now classified by WHO 2016 as a separate entity under myeloid malignancies owing to its unique clinico-pathologic nature, greater understanding of its distinct clinical course, but with some noted clinical, morphologic, and molecular similarities to AML and myelodysplastic syndrome (MDS). One of the most common molecular mutations observed by next-generation sequencing in the vast majority of patients with BPDCN has been the presence of TET2 mutations and variants. Notably, somatic missense and truncating mutations in TET2 have been reported in patients with both BPDCN and AML, yet their differential responses to similar therapeutic regimens in clinical trial testing indicates that there are likely key underlying etiologies that are yet to be determined.
Aims: We sought to investigate and identify critical differences between patients with BPDCN and AML at the molecular level, utilizing a series of advanced analyses including transcriptome microarray, serum multiplex immunoassays and cytokine analysis.
Methods: In order to discern these differences, we profiled bone marrow, peripheral blood and serum samples from primary patients samples with BPDCN (N = 16) and TET2-mutated AML (AMLTET2m) (N = 9) using 3 different assays. We first ascertained somatic point mutations and copy number alterations of 300 genes in our BPDCN specimens using an in-house hematologic malignancy panel ("T300" panel). Next, we confirmed the prevalence of compound truncating TET2 mutations in patients with BPDCN and few copy number alterations in the genes profiled. We then used the transcriptome microarray (ThermoFisher Scientific ClariomTM D Pico Assay, and serum multiplex immunoassays (Cytokine/Chemokine/Growth Factor 45-Plex Human ProcartaPlex™ Panel 1 (ThermoFisher Scientific, formerly Affymetrix) with the addition of IL-3 Human ProcartaPlex™ Simplex Kit, formerly Affymetrix) to compare BPDCN specimens against those from TET2-mutated AML patients.
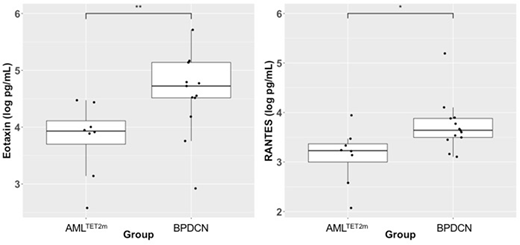
Results: With the microarray analysis, we found 920 genes to be up-regulated and 791 genes down-regulated in BPDCN specimens as compared to AMLTET2m. We corroborated known differentially expressed marker genes: higher levels of IL3Ra and TCL1A and lower levels of MPO in BPDCN as compared to AMLTET2m specimens. Genes specific to dendritic cells (PTPRS, LTK, LAMP5) were highly expressed in BPDCN than in AMLTET2m specimens. Of interest, two of these genes, PTPRS and LTK, provide possible links to the skin lesions as PTPRS is implicated in the progression of melanoma and LTK is involved in pigmentation of melanocytes. The serum cytokine profile analysis showed significantly elevated levels of eotaxin and RANTES in the BPDCN cohort as compared to the AMLTET2m cohort (Figure 1a,b). Both of these are implicated in allergic and autoimmune reactions by behaving as eosinophil chemo-attractants. Along with the higher levels of PTPRS and dendritic nature of the tumor cells, these findings suggest a possible autoimmune background which exists in the context of disease.
Conclusions: In this novel analysis, we observed elevated levels of eotaxin and RANTES in patients with BPDCN as compared to AMLTET2m. These findings may represent an important aspect of pDC functioning even outside of BPDCN, as pDCs may contribute to the pathogenesis of systemic lupus erythematosus (SLE), an autoimmune disorder with hallmark cutaneous lesions. Moreover, autoimmune pathologies have been hypothesized to damage the bone marrow and induce destruction of myeloid precursor cells. This may incorporate some of the dendritic cell nature since in its natural context, as pDCs serve to recognize foreign particles such as viruses and synthetic oligonucleotides through Toll-like Receptors TLR7/9. These findings suggest that further study into these markers are warranted in patients with BPDCN.
Figure 1. Differential serum cytokine levels between BPDCN and AMLTET2m (a) Eotaxin (pg/mL), Wilcox rank test P < 0.01 (b) RANTES (pg/mL), Wilcox rank test P < 0.05.
Konopleva:Stemline Therapeutics: Research Funding. Pemmaraju:stemline: Consultancy, Honoraria, Research Funding; plexxikon: Research Funding; SagerStrong Foundation: Research Funding; daiichi sankyo: Research Funding; celgene: Consultancy, Honoraria; Affymetrix: Research Funding; samus: Research Funding; cellectis: Research Funding; abbvie: Research Funding; novartis: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal